 © 1998 Division of Chemical Education, Inc., American Chemical Society. | Chemistry Comes Alive! (entry page) | Table of Contents | Index | Textbook Cross Reference | Frequently Asked Questions | Matrix of Topics | In the last demonstration, the three metals are lithium, sodiumand potassium. Write a balanced chemical equation for the reaction of lithiumwith water.ģ. What is the trend in the reactivity of the three alkali metalswith water?Ģ. Potassium's reaction with water is much more vigorous than sodium's.ĭemonstrationNotes: Warnings, Safety Information, etc.ġ. The aluminum pastes and stamped powder are free from the reaction with water. Theproducts of sodium's reaction with water are hydrogen gas and sodiumhydroxide, the base that turns the phenolphthalein pink. 
Note that the sodium almost immediately melts,and then skims the surface more rapidly than the lithium did. The reaction of sodium metal with water is more vigorous thanlithium's reaction. The water turns pinkas the reaction progresses because lithium hydroxide, a base, is alsoproduced. The lithium skims along theliquid surface, and hydrogen gas is generated. Small chunks of lithium, sodium and potassium react with water,generating hydrogen gas and turning phenolphthalein indicator pink.The reactivities of the three metals with water are compared.ĭescriptive chemistry, indicators, elements,reactivity, safety, evidence of chemical reaction, periodic trends,reaction rates, redox reaction, periodic table, oxidation and reduction,metals, rates of reactionĪ small chunk of lithium metal is placed in a dish of watercontaining phenolphthalein indicator.  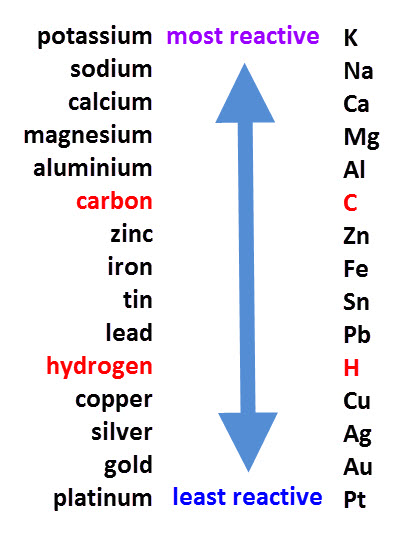
Relative Reactivity of Alkali Metals Relative Reactivity of Alkali Metals 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
